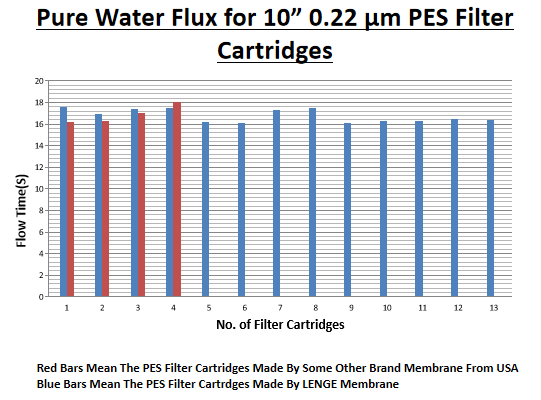
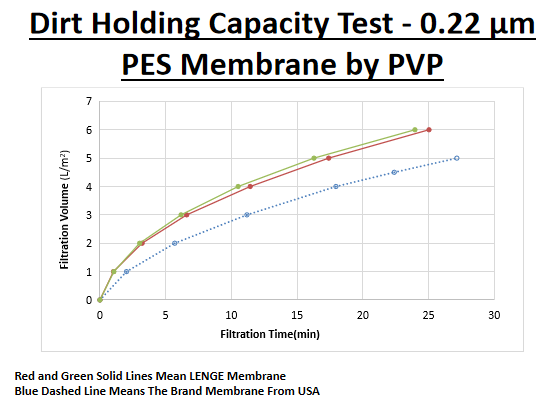
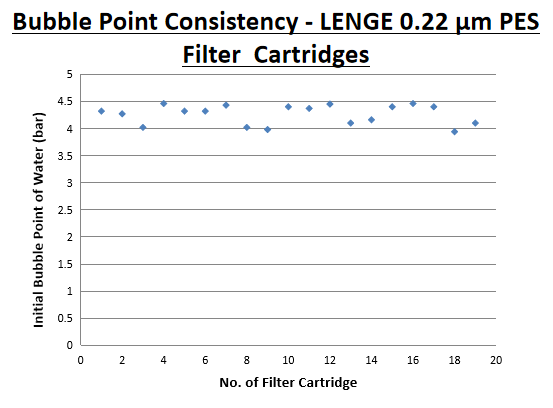
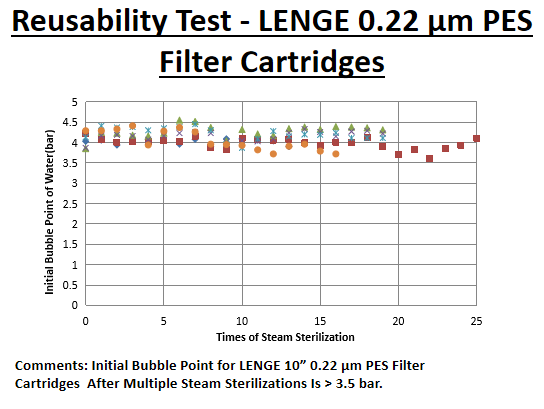
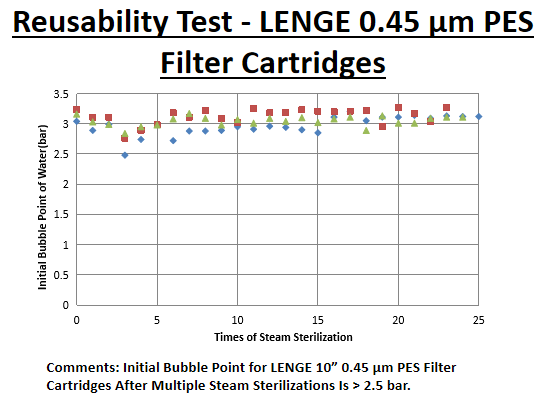
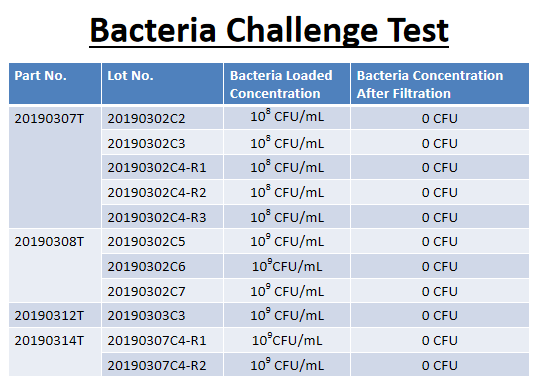
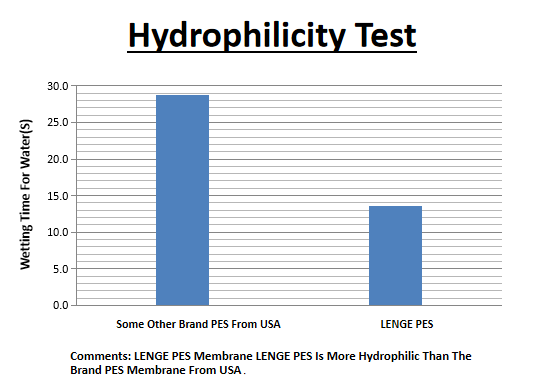
Information

X
- Home
-
Products
- Purification Equipment
- Air Filter
-
Pleated Filter Cartridge
- PES Microfiltration Membrane
- Nylon pleated filter element
- PTFE Microporous membrane pleated filter element
- PP Pleated filter element
- GF Pleated filter element
- Capsule Filter
- MS Series String Wound Filter Cartridge
- WS series string wound filter cartridges
- PTFE Sintered Tube
- Titanium Alloy Sintered Tube
- Filter Bag
- Filter Housings
- Stainless Steel Furnishing
- Why LENGE
- Solutions
- About LENGE
- News